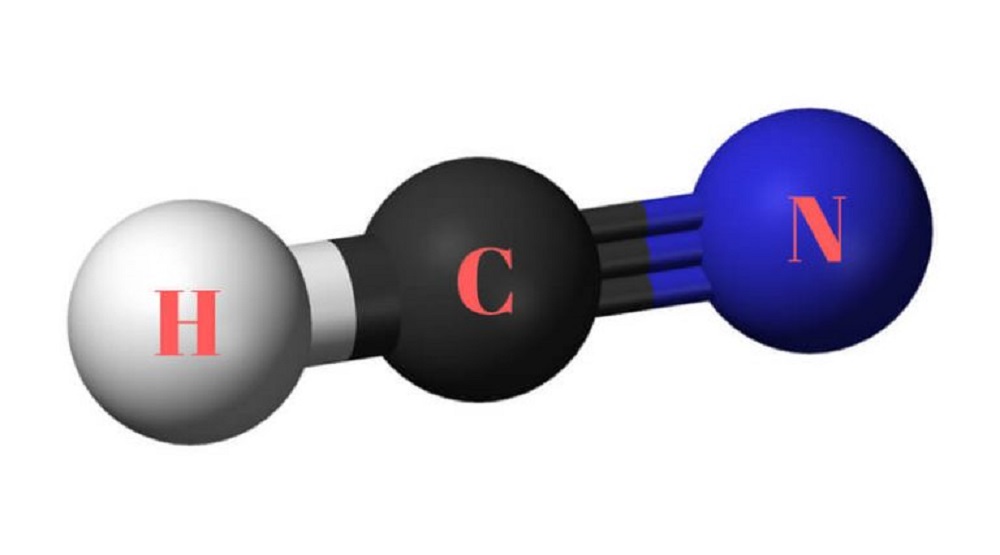
Hydrogen cyanide (HCN) is a dull, quickly acting, exceptionally toxic gas or fluid that has a smell of harsh almonds. Most HCN is utilized as a halfway at the site of creation. Significant utilizations incorporate the production of nylons, plastics, and fumigants. hydrogen cyanide.
HCN
![]() Hydrogen cyanide, furthermore called formonitrile (HCN), a significantly temperamental, dreary, and incredibly destructive liquid (limit 26° C [79° F], edge of freezing solid - 14° C [7° F]). A response of hydrogen cyanide in water is called hydrocyanic destructive, or prussic destructive.
Hydrogen cyanide, furthermore called formonitrile (HCN), a significantly temperamental, dreary, and incredibly destructive liquid (limit 26° C [79° F], edge of freezing solid - 14° C [7° F]). A response of hydrogen cyanide in water is called hydrocyanic destructive, or prussic destructive.
![]() It was viewed as in 1782 by a Swedish researcher, Carl Wilhelm Scheele, who set it up from the shade Prussian blue. Hydrogen cyanide and its combinations are used for a few, substance processes, including fumigation, the case setting of iron and steel, electroplating, and the intermingling of minerals.
It was viewed as in 1782 by a Swedish researcher, Carl Wilhelm Scheele, who set it up from the shade Prussian blue. Hydrogen cyanide and its combinations are used for a few, substance processes, including fumigation, the case setting of iron and steel, electroplating, and the intermingling of minerals.
![]() It in like manner is used in the availability of acrylonitrile, which is used in the production of acrylic fibers, made versatile, and plastics.
It in like manner is used in the availability of acrylonitrile, which is used in the production of acrylic fibers, made versatile, and plastics.
![]() Hydrogen cyanide, furthermore called form nitrile (HCN), a significantly shaky, dreary, and incredibly hurtful liquid (limit 26° C [79° F], edge of freezing solid - 14° C [7° F]). A response of hydrogen in water is called hydrocyanic destructive, or prussic destructive.
Hydrogen cyanide, furthermore called form nitrile (HCN), a significantly shaky, dreary, and incredibly hurtful liquid (limit 26° C [79° F], edge of freezing solid - 14° C [7° F]). A response of hydrogen in water is called hydrocyanic destructive, or prussic destructive.
![]() It was viewed as in 1782 by a Swedish researcher, Carl Wilhelm Scheele, who set it up from the shade Prussian blue.
It was viewed as in 1782 by a Swedish researcher, Carl Wilhelm Scheele, who set it up from the shade Prussian blue.
![]() Hydrogen cyanide and its blends are used for a few, substance processes, including fumigation, the case setting of iron and steel, electroplating, and the combination of minerals. It similarly is used in the availability of acrylonitrile, which is used in the production of acrylic fibers, made flexible, and plastics.
Hydrogen cyanide and its blends are used for a few, substance processes, including fumigation, the case setting of iron and steel, electroplating, and the combination of minerals. It similarly is used in the availability of acrylonitrile, which is used in the production of acrylic fibers, made flexible, and plastics.
![]() Hydrogen cyanide may be isolated in little sums from plants, where it occurs in blend in with sugars. Gigantic measures of hydrogen cyanide for research focus and business use are fused by three head strategies:
Hydrogen cyanide may be isolated in little sums from plants, where it occurs in blend in with sugars. Gigantic measures of hydrogen cyanide for research focus and business use are fused by three head strategies:
(1) treatment of sodium cyanide with sulfuric destructive
(2) reactant oxidation of a methane–smelling salts blend
(3) breaking down of formamide (HCONH2).
![]() Hydrogen cyanide is a brilliant dissolvable for certain salts, yet it isn’t extensively used as a dissolvable because of its destructiveness. In pure design, hydrogen cyanide is a consistent compound, but it polymerizes expeditiously inside seeing fundamental substances, such as smelling salts or sodium cyanide.
Hydrogen cyanide is a brilliant dissolvable for certain salts, yet it isn’t extensively used as a dissolvable because of its destructiveness. In pure design, hydrogen cyanide is a consistent compound, but it polymerizes expeditiously inside seeing fundamental substances, such as smelling salts or sodium cyanide.
![]() The salts are used in the extraction of metals, in electrolytic cycles, and in the treatment of steel. Among the critical reactions with normal combinations are those with aldehydes and ketones, forming cyanohydrins, which fill in as intermediates in various regular mixes, and with ethylene oxide, outlining a widely appealing thing that is changed over to acrylonitrile (CH2=CHCN).
The salts are used in the extraction of metals, in electrolytic cycles, and in the treatment of steel. Among the critical reactions with normal combinations are those with aldehydes and ketones, forming cyanohydrins, which fill in as intermediates in various regular mixes, and with ethylene oxide, outlining a widely appealing thing that is changed over to acrylonitrile (CH2=CHCN).
Carbonate
![]() Carbonate, any individual from two classes of synthetic mixtures got from carbonic corrosive or carbon dioxide (q.v.).
Carbonate, any individual from two classes of synthetic mixtures got from carbonic corrosive or carbon dioxide (q.v.).
The inorganic carbonates are salts of carbonic corrosive (H2CO3), containing the carbonate particle, CO2/3-, and particles of metals like sodium or calcium.
![]() Inorganic carbonates contain numerous minerals (see carbonate mineral) and are the chief constituents of limestones and dolomites; they additionally involve the hard aspects of numerous marine spineless creatures.
Inorganic carbonates contain numerous minerals (see carbonate mineral) and are the chief constituents of limestones and dolomites; they additionally involve the hard aspects of numerous marine spineless creatures.
Natural carbonates are esters; that is, compounds in which the hydrogen molecules of carbonic corrosive have been supplanted via carbon-containing consolidating gatherings like ethyl, C2H5.
Arrhenius hypothesis
![]() Arrhenius hypothesis, hypothesis, presented in 1887 by the Swedish researcher Svante Arrhenius, that acids are substances that separate in water to yield electrically charged iotas or atoms, called particles, one of which is a hydrogen particle (H+), and that bases ionize in water to yield hydroxide particles (OH−).
Arrhenius hypothesis, hypothesis, presented in 1887 by the Swedish researcher Svante Arrhenius, that acids are substances that separate in water to yield electrically charged iotas or atoms, called particles, one of which is a hydrogen particle (H+), and that bases ionize in water to yield hydroxide particles (OH−).
![]() It is currently realized that the hydrogen particle can’t exist alone in water arrangement; rather, it exists in a consolidated state with a water atom, as the hydronium particle (H3O+).
It is currently realized that the hydrogen particle can’t exist alone in water arrangement; rather, it exists in a consolidated state with a water atom, as the hydronium particle (H3O+).
Practically speaking the hydronium particle is still usually alluded to as the hydrogen particle.
![]() The acidic conduct of some notable acids (e.g., sulfuric, hydrochloric, nitric, and acidic acids) and the essential properties of notable hydroxides (e.g., sodium, potassium, and calcium hydroxides) are clarified as far as their capacity to yield hydrogen and hydroxide particles, individually, in arrangement.
The acidic conduct of some notable acids (e.g., sulfuric, hydrochloric, nitric, and acidic acids) and the essential properties of notable hydroxides (e.g., sodium, potassium, and calcium hydroxides) are clarified as far as their capacity to yield hydrogen and hydroxide particles, individually, in arrangement.
Moreover, such acids and bases might be named solid or powerless acids and bases relying upon the hydrogen particle or hydroxide particle fixation created in arrangement.
The response between a corrosive and a base prompts the arrangement of a salt and water; the last option is the consequence of the blend of a hydrogen particle and a hydroxide particle.
Science Imagine
![]() Science, any arrangement of information that is worried about the actual world and its peculiarities and that involves fair perceptions and precise experimentation. As a rule, a science includes a quest for information covering general facts or the tasks of central laws.
Science, any arrangement of information that is worried about the actual world and its peculiarities and that involves fair perceptions and precise experimentation. As a rule, a science includes a quest for information covering general facts or the tasks of central laws.
![]() Science can be partitioned into various branches dependent regarding the matter of study. The actual sciences concentrate on the inorganic world and involve the fields of cosmology, physical science, science, and the Studies of the planet.
Science can be partitioned into various branches dependent regarding the matter of study. The actual sciences concentrate on the inorganic world and involve the fields of cosmology, physical science, science, and the Studies of the planet.
The natural sciences, for example, science and medication concentrate on the natural universe of life and its cycles. Sociologies like humanities and financial matters concentrate on the social and social parts of human conduct.
![]() Science is additionally treated in various articles. For the historical backdrop of Western and Eastern science, see science, history of. For the conceptualization of science and its interrelationships with culture, see science, theory of. For the essential parts of the logical methodology, see actual science, standards of; and logical technique.
Science is additionally treated in various articles. For the historical backdrop of Western and Eastern science, see science, history of. For the conceptualization of science and its interrelationships with culture, see science, theory of. For the essential parts of the logical methodology, see actual science, standards of; and logical technique.
Phosphate
![]() Phosphate, any of various substance intensifies identified with phosphoric corrosive (H3PO4). One gathering of these subordinates is made out of salts containing the phosphate particle (PO43−),.
Phosphate, any of various substance intensifies identified with phosphoric corrosive (H3PO4). One gathering of these subordinates is made out of salts containing the phosphate particle (PO43−),.
![]() The hydrogen phosphate particle (HPO42−), or the dihydrogen phosphate particle (H2PO4−), and emphatically charged particles like those of sodium or calcium; a subsequent gathering is made out of esters, in which the hydrogen iotas of phosphoric corrosive have been supplanted by natural consolidating gatherings like ethyl (C2H5) or phenyl (C6H5).
The hydrogen phosphate particle (HPO42−), or the dihydrogen phosphate particle (H2PO4−), and emphatically charged particles like those of sodium or calcium; a subsequent gathering is made out of esters, in which the hydrogen iotas of phosphoric corrosive have been supplanted by natural consolidating gatherings like ethyl (C2H5) or phenyl (C6H5).
Phosphoric corrosive
![]() Phosphoric corrosive, likewise called orthophosphoric corrosive, (H3PO4), the main oxygen corrosive of phosphorus, used to make phosphate salts for composts.
Phosphoric corrosive, likewise called orthophosphoric corrosive, (H3PO4), the main oxygen corrosive of phosphorus, used to make phosphate salts for composts.
It is likewise utilized in dental concretes, in the readiness of egg whites subsidiaries, and in the sugar and material businesses. It fills in as an acidic, fruitlike enhancing in food items.
![]() Unadulterated phosphoric corrosive is a translucent strong (liquefying point 42.35° C, or 108.2° F); in less thought structure it is a dry sweet fluid. The unrefined corrosive is ready from phosphate rock, while corrosive of higher virtue is produced using white phosphorus.
Unadulterated phosphoric corrosive is a translucent strong (liquefying point 42.35° C, or 108.2° F); in less thought structure it is a dry sweet fluid. The unrefined corrosive is ready from phosphate rock, while corrosive of higher virtue is produced using white phosphorus.
![]() Phosphoric corrosive structures three classes of salts comparing to substitution of one, two, or three hydrogen iotas. Among the significant phosphate salts are:
Phosphoric corrosive structures three classes of salts comparing to substitution of one, two, or three hydrogen iotas. Among the significant phosphate salts are:
sodium dihydrogen phosphate (NaH2PO4), utilized for control of hydrogen particle fixation (acridity) of arrangements; disodium hydrogen phosphate (Na2HPO4), utilized in water treatment as a precipitant for profoundly charged metal cations; trisodium phosphate (Na3PO4).
Utilized in cleansers and cleansers; calcium dihydrogen phosphate or calcium superphosphate (Ca[H2PO4]2), a significant manure fixing; calcium monohydrogen phosphate (CaHPO4), utilized as a molding specialist for salts and sugars.
![]() Phosphoric corrosive atoms communicate under reasonable conditions, regularly at high temperatures, to shape bigger particles (ordinarily with loss of water). In this way, diphosphoric, or pyrophosphoric, corrosive (H4P2O7) is framed from two particles of phosphoric corrosive, less one atom of water.
Phosphoric corrosive atoms communicate under reasonable conditions, regularly at high temperatures, to shape bigger particles (ordinarily with loss of water). In this way, diphosphoric, or pyrophosphoric, corrosive (H4P2O7) is framed from two particles of phosphoric corrosive, less one atom of water.
It is the most straightforward of a homologous series of long chain atoms called polyphosphoric acids, with the overall equation H(HPO3)nOH, where n = 2, 3, 4, . . . . Metaphosphoric acids, (HPO3)n, in which n = 3, 4, 5, . . ., are one more class of polymeric phosphoric acids. The known metaphosphoric acids are portrayed by cyclic atomic designs.
The term metaphosphoric corrosive is utilized likewise to allude to a gooey, tacky substance that is a combination of both long chain and ring types of (HPO3)n. The different polymeric types of phosphoric corrosive are additionally ready by hydration of phosphorus oxides.
Carbonic acid
![]() Carbonic corrosive, (H2CO3), a compound of the components hydrogen, carbon, and oxygen. It is framed in modest quantities when its anhydride, carbon dioxide (CO2), breaks up in water.
Carbonic corrosive, (H2CO3), a compound of the components hydrogen, carbon, and oxygen. It is framed in modest quantities when its anhydride, carbon dioxide (CO2), breaks up in water.
![]() CO2 + H2O ⇌ H2CO3The dominating species are essentially approximately hydrated CO2 particles. Carbonic corrosive can be viewed as a diprotic corrosive from which two series of salts can be framed—to be specific, hydrogen carbonates, containing HCO3−, and carbonates, containing CO32−.H2CO3 + H2O ⇌ H3O+ + HCO3−
CO2 + H2O ⇌ H2CO3The dominating species are essentially approximately hydrated CO2 particles. Carbonic corrosive can be viewed as a diprotic corrosive from which two series of salts can be framed—to be specific, hydrogen carbonates, containing HCO3−, and carbonates, containing CO32−.H2CO3 + H2O ⇌ H3O+ + HCO3−
-
HCO3− + H2O ⇌ H3O+ + CO32−However, the corrosive base conduct of carbonic corrosive relies upon the various paces of a portion of the responses in question, just as their reliance on the pH of the framework. For instance, at a pH of under 8, the essential responses and their general speed are as follows:CO2 + H2O ⇌ H2CO3 (slow)
-
H2CO3 + OH− ⇌ HCO3− + H2O (fast)Above pH 10 the accompanying responses are important:CO2 + OH− ⇌ HCO3− (slow)
-
HCO3− + OH− ⇌ CO32− + H2O (fast)Between pH upsides of 8 and 10, all the above harmony responses are huge.
![]() Carbonic corrosive assumes a part in the gathering of caverns and cavern developments like cave rock formations and stalagmites. The biggest and most normal caverns are those shaped by disintegration of limestone or dolomite by the activity of water rich in carbonic corrosive got from ongoing precipitation.
Carbonic corrosive assumes a part in the gathering of caverns and cavern developments like cave rock formations and stalagmites. The biggest and most normal caverns are those shaped by disintegration of limestone or dolomite by the activity of water rich in carbonic corrosive got from ongoing precipitation.
![]() The calcite in tapered rocks and stalagmites is gotten from the overlying limestone close to the bedrock/soil interface.
The calcite in tapered rocks and stalagmites is gotten from the overlying limestone close to the bedrock/soil interface.
Water penetrating through the dirt ingests carbon dioxide from the carbon dioxide-rich soil and structures a weaken arrangement of carbonic corrosive. At the point when this corrosive water arrives at the foundation of the dirt, it responds with the calcite in the limestone bedrock and brings some of it into arrangement.
![]() The water proceeds with its descending course through tight joints and cracks in the unsaturated zone with minimal further substance response.
The water proceeds with its descending course through tight joints and cracks in the unsaturated zone with minimal further substance response.
At the point when the water rises out of the cavern rooftop, carbon dioxide is lost into the cavern environment, and a portion of the calcium carbonate is hastened.
The penetrating water goes about as a calcite siphon, eliminating it from the highest point of the bedrock and redepositing it in the cavern underneath.
![]() Carbonic corrosive is significant in the vehicle of carbon dioxide in the blood. Carbon dioxide enters blood in the tissues since its nearby fractional tension is more prominent than its incomplete strain in blood moving through the tissues.
Carbonic corrosive is significant in the vehicle of carbon dioxide in the blood. Carbon dioxide enters blood in the tissues since its nearby fractional tension is more prominent than its incomplete strain in blood moving through the tissues.
As carbon dioxide enters the blood, it consolidates with water to frame carbonic corrosive, which separates into hydrogen particles (H+) and bicarbonate particles (HCO3-).
Blood corrosiveness is negligibly impacted by the delivered hydrogen particles since blood proteins, particularly hemoglobin, are compelling buffering specialists.
A cushion arrangement opposes change in sharpness by joining with added hydrogen particles and, basically, inactivating them.
![]() The regular transformation of carbon dioxide to carbonic corrosive is a somewhat lethargic cycle; notwithstanding, carbonic anhydrase, a protein compound present inside the red platelet, catalyzes this response with adequate quickness that it is refined in just a small portion of a second.
The regular transformation of carbon dioxide to carbonic corrosive is a somewhat lethargic cycle; notwithstanding, carbonic anhydrase, a protein compound present inside the red platelet, catalyzes this response with adequate quickness that it is refined in just a small portion of a second.
![]() Since the chemical is available just inside the red platelet, bicarbonate gathers to a lot more prominent degree inside the red cell than in the plasma.
Since the chemical is available just inside the red platelet, bicarbonate gathers to a lot more prominent degree inside the red cell than in the plasma.
![]() The limit of blood to convey carbon dioxide as bicarbonate is improved by a particle transport framework inside the red platelet layer that at the same time moves a bicarbonate particle out of the cell and into the plasma in return for a chloride particle.
The limit of blood to convey carbon dioxide as bicarbonate is improved by a particle transport framework inside the red platelet layer that at the same time moves a bicarbonate particle out of the cell and into the plasma in return for a chloride particle.
![]() The concurrent trade of these two particles, known as the chloride shift, allows the plasma to be utilized as a capacity site for bicarbonate without changing the electrical charge of either the plasma or the red platelet.
The concurrent trade of these two particles, known as the chloride shift, allows the plasma to be utilized as a capacity site for bicarbonate without changing the electrical charge of either the plasma or the red platelet.
![]() Just 26% of the all out carbon dioxide content of blood exists as bicarbonate inside the red platelet, while 62% exists as bicarbonate in plasma; be that as it may, the greater part of bicarbonate particles is first created inside the cell, then, at that point, shipped to the plasma.
Just 26% of the all out carbon dioxide content of blood exists as bicarbonate inside the red platelet, while 62% exists as bicarbonate in plasma; be that as it may, the greater part of bicarbonate particles is first created inside the cell, then, at that point, shipped to the plasma.
![]() An opposite arrangement of responses happens when blood arrives at the lung, where the incomplete strain of carbon dioxide is lower than in the blood.
An opposite arrangement of responses happens when blood arrives at the lung, where the incomplete strain of carbon dioxide is lower than in the blood.
Nitrous corrosive
Nitrous corrosive, (HNO2), an unsound, pitifully acidic compound that has been arranged distinctly as cool, weaken arrangements.
It is helpful in science in changing over amines into diazonium compounds, which are utilized in making azo colors. It is typically ready by acidifying an answer of one of its salts, the nitrites, which are more steady (see nitrite).
Nitrous corrosive deteriorates into nitric oxide, NO, and nitric corrosive, HNO3. It might respond as either an oxidizing or a diminishing specialist; that is, its nitrogen iota may either acquire or lose electrons in responses with different substances.
Nitrous corrosive, for instance, oxidizes iodide particle to basic iodine yet diminishes bromine to bromide particle.
Rochelle salt, additionally called Sodium Potassium Tartrate Tetrahydrate, a translucent strong having a huge piezoelectric impact (electric charge prompted on its surfaces by mechanical distortion because of strain, contorting, or twisting), making it valuable in delicate acoustical and vibrational gadgets.
Like other piezoelectric materials, Rochelle salt gems (KNaC4H4O6·4H2O) become stressed when exposed to electric fields. They decay at tolerably high temperatures (55° C [131° F]) and require insurance against dampness.
Piezoelectric twisting is straightforwardly corresponding to the applied electric field and turns around as the extremity of the applied field is switched.
These fundamental properties are put to use in electromechanical transducers like ultrasonic generators, mouthpieces, and phonograph pickups and in electromechanical resonators. See likewise tartaric corrosive.
Ammonium chloride
Ammonium chloride (NH4Cl), additionally called sal ammoniac, the salt of alkali and hydrogen chloride.
Its chief uses are as a nitrogen supply in manures and as an electrolyte in dry cells, and it is additionally broadly utilized as a constituent of arousing, tinning, and patching motions to eliminate oxide coatings from metals and in this way work on the attachment of the welds.
It is a part of numerous exclusive virus drugs and hack cures in view of its viability as an expectorant, and in veterinary medication, it is utilized to forestall urinary stones in goats, steers, and sheep. Ammonium chloride is a dismal glasslike substance.
It is exceptionally dissolvable in water, promptly framing a somewhat acidic arrangement. It disintegrates without dissolving at 340 °C (644 °F) to shape equivalent volumes of smelling salts and hydrogen chloride.
Ammonium chloride is yielded as a result in the smelling salts soft drink process for making sodium carbonate.
It additionally is delivered by response of ammonium sulfate and sodium chloride arrangements. When blended in with slaked lime (calcium carbonate), alkali gas is the outcome.
Hydrogen cyanide truth sheet
Otherwise called hydrocyanic corrosive (CAS Library Number 74‐90‐8) or HCN, hydrogen cyanide is a rapid‐acting deadly specialist that represses high-impact breath at the phone level, keeping cells from using oxygen.
HCN, which at climatic strain happens over the temperature range - 14 °C to +26 °C, is lackluster gas or somewhat blue white fluid.
On standing, it polymerizes and may detonate, however it tends to be balanced out. Certain individuals can smell HCN at low fixations, portraying a fragrance of harsh almonds or marzipan; others can’t recognize it.
Operations
Inward breath is the most probable course of passage, causing hyperventilation at first. HCN fume doesn’t cross skin. A hydrogen cyanide centralization of 300 mg/m3 in air will kill a human inside around 10 minutes. Fluid HCN will enter skin or can be ingested from the lungs, when scattered as a spray.
Idleness period and recuperation time
Manifestations of harming are fast in beginning since it is immediately assimilated from the lungs. Hyperventilation happens first and increments with the portion breathed in (relies upon season of openness and fixation). This is trailed by quick loss of awareness at high focuses.
-
Primary clinical side effects
-
At high focuses
-
Hyperventilation
-
Loss of awareness
-
Seizures
-
Loss of corneal reflex
-
Vibe of throat choking
-
Happiness
-
Disarray
-
Disabled vision
-
Sensation of tightening around the head
-
Agony might happen toward the rear of the neck and chest
-
Quick and moderate feeling of warmth (because of vasodilation) with noticeable flushing
-
Surrender follows with sickness, retching, cerebral pain, trouble in breathing and a sensation of snugness around the chest
-
Obviousness and asphyxia are unavoidable except if openness stops
-
Apprehensiveness
-
Dyspnea
-
Cerebral pain
-
Dizziness
Notice a metallic desire for the mouth
![]() Distinctive degree of openness implies that those showing up on the scene will observe setbacks who are: a symptomatic; showing intense indications; recuperating from them. The patients ought to be taken out from the wellspring of openness. Emergency ought to be performed.
Distinctive degree of openness implies that those showing up on the scene will observe setbacks who are: a symptomatic; showing intense indications; recuperating from them. The patients ought to be taken out from the wellspring of openness. Emergency ought to be performed.
![]() Casualties who are asymptomatic a few minutes later openness don’t need oxygen or counteractants.
Casualties who are asymptomatic a few minutes later openness don’t need oxygen or counteractants.
![]() Where openness has caused intense impacts (spasms, apnea), oxygen (preferably 100%)and cures ought to be controlled right away.
Where openness has caused intense impacts (spasms, apnea), oxygen (preferably 100%)and cures ought to be controlled right away.
:small_blue_diamond:Patients recuperating from intense openings (and oblivious, yet breathing) will make a quicker recuperation with remedys and oxygen.
![]() Assets allowing, revival ought to be endeavored on subjects with no heartbeat on the off chance that heart stoppage is later.
Assets allowing, revival ought to be endeavored on subjects with no heartbeat on the off chance that heart stoppage is later.
![]() Assuming the openness was to cyanide gas, sterilization of apparel or hardware is superfluous taking into account its high unpredictability. Fluid pollution will require purification with water and cleanser.
Assuming the openness was to cyanide gas, sterilization of apparel or hardware is superfluous taking into account its high unpredictability. Fluid pollution will require purification with water and cleanser.
![]() Treatment should be expeditious. Later oxygen has been managed, resulting treatment is pointed toward separating the cyanide particle into cytochrome oxidase. Treatments might incorporate (all treatment is to be utilized under a doctor’s heading, for suggestive patient/casualties):
Treatment should be expeditious. Later oxygen has been managed, resulting treatment is pointed toward separating the cyanide particle into cytochrome oxidase. Treatments might incorporate (all treatment is to be utilized under a doctor’s heading, for suggestive patient/casualties):
Routes of Exposure
Inhalation
Hydrogen cyanide is promptly retained from the lungs; indications of harming start inside the space of seconds to minutes.
The scent of hydrogen cyanide is noticeable at 2-10 ppm (OSHA PEL = 10 ppm), however doesn’t give satisfactory admonition of perilous fixations.
View of the scent is a hereditary quality (20% to 40% of everybody can’t distinguish hydrogen cyanide); likewise, quick olfactory weariness can happen. Hydrogen cyanide is lighter than air.
Youngsters presented to similar degrees of hydrogen cyanide as grown-ups may get bigger portions since they have more noteworthy lung surface area:body weight proportions and expanded moment volumes:weight proportions.
Skin/Eye to eye connection
Openness to hydrogen cyanide can cause skin and eye bothering. All the more critically, skin or eye assimilation is quick and adds to foundational harming. Later skin openness, beginning of indications might be prompt or postponed for 30 to an hour.
Most instances of harmfulness from dermal openness have been from modern mishaps including halfway inundation in fluid cyanide or cyanide arrangements or from contact with liquid cyanide salts, bringing about enormous surface-region consumes.
Youngsters are more helpless against poisons retained through the skin due to their moderately bigger surface area:body weight proportion.
Ingestion
Ingestion of hydrogen cyanide arrangements or cyanide salts can be quickly lethal.
Sources/Employments
Hydrogen cyanide is fabricated by oxidation of alkali methane combinations under controlled conditions and by the reactant deterioration of formamide.
It could be created by treating cyanide salts with corrosive, and it is an ignition side-effect of nitrogen-containing materials like fleece, silk, and plastics. It is additionally created by enzymatic hydrolysis of nitriles and related synthetic substances. Hydrogen cyanide gas is a result of coke-stove and impact heater activities.
Hydrogen cyanide is utilized in disinfecting; electroplating; mining; and in delivering manufactured filaments, plastics, colors, and pesticides. It additionally is utilized as a halfway in substance combinations.
Principles and Rules
-
OSHA PEL (allowable openness limit) (roof) = 10 ppm (skin) (arrived at the midpoint of north of 15 minutes)
-
NIOSH IDLH (promptly risky to life or wellbeing) = 50 ppm
AIHA ERPG-2 (crisis reaction arranging rule) (most extreme airborne fixation underneath which it is accepted that practically everything people could be presented for as long as 1 hour without encountering or creating irreversible or other genuine wellbeing impacts or manifestations which could weaken a singular’s capacity to make a defensive move) = 10 ppm
Actual Properties
-
Dismal gas or boring or light blue fluid
-
Cautioning properties: Almond scent at >l ppm; insufficient admonition for intense or ongoing openness
-
Sub-atomic weight: 27.03 daltons
-
Limit (760 mm Hg): 78ºF (25.6ºC)
-
Edge of freezing over: 8ºF (- 13.4ºC)
-
Explicit gravity (fluid): 0.69 (water = 1)
-
Fume pressure: 630 mm Hg at 68ºF (20ºC)
-
Gas thickness: 0.94 (air = 1)
-
Water solvency: Combustible at
-
Temperatures >0ºF (- 18ºC)
-
Combustibility: combustible limits 3.9% to 21.8% at room temperature
-
Combustible reach: 5.6% to 40% (fixation in air)
Contradictions
Hydrogen cyanide responds with amines, oxidizers, acids, sodium hydroxide, calcium hydroxide, sodium carbonate, scathing substances, and smelling salts. Hydrogen cyanide might polymerize at 122ºF to 140ºF.
Health Effects
Hydrogen cyanide is exceptionally harmful by all courses of openness and may cause unexpected beginning of significant CNS, cardiovascular, and respiratory impacts, prompting demise in practically no time.
Exposure to bring down groupings of hydrogen cyanide might create eye disturbance, cerebral pain, disarray, queasiness, and heaving continued at times by unconsciousness and demise.
Hydrogen cyanide goes about as a cell asphyxiant. By restricting to mitochondrial cytochrome oxidase, it forestalls the usage of oxygen in cell digestion. The CNS and myocardium are especially delicate to the poisonous impacts of cyanide.
Acute Exposure
In people, cyanide joins with the ferric particle in mitochondrial cytochrome oxidase, forestalling electron transport in the cytochrome framework and stopping oxidative phosphorylation and ATP creation.
The restraint of oxidative digestion puts expanded requests on anaerobic glycolysis, which brings about lactic corrosive creation and may deliver extreme corrosive base awkwardness. The CNS is especially touchy to the poisonous impacts of cyanide, and openness to hydrogen cyanide for the most part creates indications inside a brief timeframe.
Youngsters don’t generally react to synthetic substances similarly that grown-ups do. Various conventions for dealing with their consideration might be required.
CNS
CNS signs and manifestations typically grow quickly. Introductory indications are vague and incorporate fervor, discombobulation, sickness, regurgitating, migraine, and shortcoming.
As harming advances, languor, tetanic fit, jaw spasming, seizures, visualizations, loss of awareness, and unconsciousness might happen.
Cardiovascular
Strange heartbeat can happen in instances of serious harming. Slow heartbeat, immovable low circulatory strain, and demise might result. Hypertension and a fast heartbeat might be early, transient discoveries.
Respiratory
Later fundamental harming starts, casualties might gripe of windedness and chest snugness. Aspiratory discoveries might incorporate quick breathing and expanded profundity of breaths.
As harming advances, breaths become slow and wheezing; a somewhat blue skin tone could possibly be available.
Gathering of liquid in the lungs might create.
Youngsters might be more defenseless against gas openness in light of moderately expanded moment ventilation per kg and inability to empty a region expeditiously when uncovered.
Metabolic
An anion-hole, metabolic acidosis happens in serious harming from expanded blood levels of lactic corrosive.
Due to their higher metabolic rates, youngsters might be more defenseless against poisons meddling with essential digestion.
Dermal
Dermal contact with hydrogen cyanide can cause skin aggravation.
Dermal retention can happen, prompting fundamental harmfulness. Retention happens all the more promptly at high surrounding temperature and relative moistness.
Due to their generally bigger surface area:body weight proportion, youngsters are more defenseless against poisons ingested through the skin.
Visual/Ophthalmic
When sprinkled in the eye, hydrogen cyanide can cause eye aggravation and expanding. Eye to eye connection with cyanide salts has created foundational manifestations in trial creatures. Widened understudies are normal in serious harming. Transient visual impairment can happen, despite the fact that it seldom does.
Expected Sequelae
Overcomers of serious openness might endure mind harm because of an immediate activity on neurons, or to absence of oxygen, or conceivably because of deficient blood dissemination.
Instances of neurologic sequelae, for example, character changes, memory shortfalls, aggravations in deliberate muscle developments, and the presence of compulsory developments (i.e., extrapyramidal disorders) have been accounted for.
Ongoing Exposure
Persistently uncovered specialists might grumble of migraine, eye bothering, simple weariness, chest distress, palpitations, loss of hunger, and nosebleeds.
Persistent openness might be more genuine for kids in view of their potential longer life expectancy.
Cancer-causing nature
Hydrogen cyanide has not been arranged for cancer-causing impacts, and no cancer-causing impacts have been accounted for hydrogen cyanide.
Regenerative and Formative Impacts
No regenerative or formative impacts of hydrogen cyanide have been accounted for in test creatures or people. Hydrogen cyanide is excluded from Regenerative and Formative Poisons, a 1991 report distributed by the U.S.
General Bookkeeping Office (GAO) that rundowns 30 synthetic substances of concern due to broadly recognized conceptive and formative results.
Expanded degrees of thiocyanate in the umbilical ropes of hatchlings whose moms smoked contrasted with those whose moms were non-smokers recommends that thiocyanate, and perhaps at the same time cyanide, can cross the placenta. No information were found relating to hydrogen cyanide in milk.
Prehospital The executives
Casualties presented uniquely to hydrogen cyanide gas don’t present optional tainting dangers to heros, however don’t endeavor revival without an obstruction.
Casualties whose apparel or skin is polluted with hydrogen cyanide fluid or arrangement can optionally taint reaction faculty by direct contact or through off-gassing fume.
Stay away from dermal contact with cyanide-sullied casualties or with gastric substance of casualties who might have ingested cyanide-containing materials.
Hydrogen cyanide harming is set apart by sudden beginning of significant poisonous impacts that might incorporate syncope, seizures, trance like state, heaving breaths, and cardiovascular breakdown, causing demise in practically no time. These impacts can happen from all courses of openness.
Casualties presented to hydrogen cyanide require steady consideration and quick organization of explicit counteractants.
Hot Zone
Heros ought to be prepared and attired prior to entering the Hot Zone.
On the off chance that the appropriate gear isn’t accessible, or then again assuming heros have not been prepared in its utilization, help ought to be gotten from a nearby or provincial Hazardous materials group or other appropriately prepared reaction association.
Hero Security
Hydrogen cyanide is a profoundly harmful foundational poison that is consumed well by inward breath and through the skin.
Respiratory Security: Positive-pressure, independent breathing contraption (SCBA) is suggested accordingly circumstances that include openness to possibly perilous degrees of hydrogen cyanide.
Skin Assurance: Substance defensive dress is suggested in light of the fact that both hydrogen cyanide fume and fluid can be ingested through the skin to create foundational poisonousness.
ABC Updates
Right away access for a patent aviation route, guarantee sufficient breath and heartbeat. In the event that injury is suspected, keep up with cervical immobilization physically and apply a cervical collar and a backboard when plausible.
Casualty Evacuation
On the off chance that casualties can walk, lead them out of the Hot Zone to the Purification Zone. Casualties who can’t walk might be eliminated on backboards or carts; in the event that these are not accessible, cautiously convey or drag casualties to security.
Think about administration of synthetically polluted kids, for example, measures to diminish fear of abandonment a youngster is isolated from a parent or other grown-up.
Disinfecting Zone
Patients presented distinctly to hydrogen cyanide gas who have no eye aggravation needn’t bother with disinfecting. They might be moved quickly to the Help Zone. Different patients will require disinfecting as depicted beneath.
Hero Insurance
Assuming openness not really settled to be protected, disinfecting might be directed by staff wearing a lower level of insurance than that ragged in the Hot Zone (portrayed previously). Nonetheless, don’t endeavor revival without a hindrance.
ABC Updates
Straightaway access for a patent aviation route, guarantee sufficient breath and heartbeat. Settle the cervical spine with a collar and a backboard assuming that injury is suspected. Control supplemental oxygen as required. Help ventilation with a sack valve-cover gadget if important.
Essential Cleaning
Speed is basic. For indicative casualties, furnish treatment with 100% oxygen and explicit remedys on a case by case basis. Treatment ought to be given all the while with sterilization systems. (For treatment, see ABC Updates, Progressed Treatment, and Remedys underneath).
Casualties who are capable may help with their own sterilization. Quickly take off debased dress while flushing uncovered skin and hair with plain water for 2 to 3 minutes, then, at that point, wash twice with gentle cleanser. Flush completely with water. Twofold pack polluted attire and individual assets. Use alert to stay away from hypothermia
while purifying youngsters or the older. Use covers or warmers when proper.
Inundate uncovered or bothered eyes with plain water or saline for 5 minutes. Proceed with eye water system during other essential consideration or transport. Eliminate contact focal points if effectively removable without extra injury to the eye.
In instances of ingestion, don’t initiate emesis. Assuming the casualty is ready, asymptomatic, and has a gag reflex, regulate a slurry of initiated charcoal (oversee at 1 gm/kg, regular grown-up portion 60-90 g, kid portion 25-50 g). A soft drink can and a straw might be of help when offering charcoal to a kid. In the event that the casualty is indicative, promptly establishment crisis life support measures including the utilization of the cyanide antitoxin unit (see Remedys beneath).
Think about proper administration of artificially polluted youngsters, for example, measures to lessen fear of abandonment a kid is isolated from a parent or other grown-up. On the off chance that conceivable, look for help from a youngster division master.
Move to Help Zone
When fundamental purification is finished, move the casualty to the Help Zone.
Support Zone
Be sure that casualties have been sterilized appropriately (see Disinfecting Zone above). Casualties who have been sterilized or who have been presented distinctly to fume by and large represent no genuine dangers of optional defilement to heros. In such cases, Backing Zone faculty require no particular defensive stuff.
ABC Updates
As soon as possible access for a patent aviation route. Assuming that injury is suspected, keep up with cervical immobilization physically and apply a cervical collar and a backboard when practical. Guarantee satisfactory breath and heartbeat. Manage supplemental oxygen as required and set up intravenous access if vital. Place on a cardiovascular screen.
Patients who quickly recover awareness and who have no different signs or manifestations may not need antitoxin treatment. The individuals who stay insensible or foster shock ought to be dealt with immediately with the remedy’s in the cyanide antitoxin unit (see Counteract ant
##Additional Decontamination.
Continue irrigating exposed skin and eyes, as appropriate.
In instances of ingestion, don’t actuate emesis. On the off chance that enacted charcoal has not been controlled already, and the casualty is ready, asymptomatic, and has a gag reflex, direct a slurry of initiated charcoal (oversee at 1 gm/kg, common grown-up portion 60-90 g, youngster portion 25-50 g).
A soft drink can and a straw might be of help when offering charcoal to a kid. Assuming the patient is indicative, promptly establishment crisis life support measures, including the utilization of a cyanide cure unit (see Remedy’s beneath).
Progressed Treatment
In instances of respiratory trade off secure aviation route and breath through endotracheal intubation. If impractical, perform if prepared and prepared to do as such.
Patients who are in stun or have seizures ought to be treated by cutting edge life support (ALS) conventions. These patients or the individuals who have arrhythmias might be truly acidotic; think about giving, under clinical watch, every persistent 1 mEq/kg intravenous sodium bicarbonate.
Remedies
- Whenever the situation allows, therapy with cyanide cures ought to be given under clinical watch to oblivious casualties who have known or emphatically speculated cyanide harming.
Cyanide counteractants amyl nitrite perles and intravenous imbuements of sodium nitrite and sodium thiosulfate-are bundled in the cyanide cure unit.
- Amyl nitrite perles ought to be broken onto a cloth cushion and held under the nose, over the Ambu-valve consumption, or put under the lip of the facial covering.
Breathe in for 30 seconds consistently and utilize a new perle like clockwork assuming sodium nitrite mixtures will be deferred.
- Assuming the patient has not reacted to oxygen and amyl nitrite treatment, mix sodium nitrite intravenously quickly.
The typical grown-up portion is 10 mL of a 3% arrangement (300 mg) imbued over definitely something like 5 minutes; the normal pediatric portion is 0.12 to 0.33 mL/kg body weight up to 10 mL mixed as above.
Screen circulatory strain during sodium nitrite organization, and slow the pace of mixture assuming hypotension creates.
- Then, imbue sodium thiosulfate intravenously. The standard grown-up portion is 50 mL of a 25% arrangement (12.5 g) imbued more than 10 to 20 minutes; the normal pediatric portion is 1.65 mL/kg of a 25% arrangement.
Rehash one-half of the underlying portion 30 minutes after the fact assuming that there is an insufficient clinical reaction.
Transport to Clinical Office
-
Just disinfected patients or patients not needing cleaning ought to be moved to a clinical office. “Body sacks” are not suggested.
-
Report to the base station and the getting clinical office the state of the patient, therapy given, and assessed season of landing in the clinical office.
-
On the off chance that a cyanide-containing arrangement has been ingested, set up the emergency vehicle in the event that the casualty heaves poisonous material. Have prepared a few towels and open plastic packs to rapidly tidy up and disengage vomitus.
Multi-Loss Emergency
Talk with the base station doctor or the local toxin control place for counsel in regards to emergency of various casualties.
Patients with proof of critical hydrogen cyanide openness, and all patients who have hydrogen cyanide ingestion ought to be shipped to a clinical office for assessment.
Patients who have just short inward breath openness and gentle or transient indications might be released from the scene later their names, locations, and phone numbers are recorded.
They ought to be encouraged to look for clinical consideration immediately assuming indications create or repeat (see Patient Data Sheet underneath).
Crisis Division The executives
Clinic work force in an encased region can be optionally debased by fume off-gassing from intensely drenched apparel or skin, or from harmful vomitus.
Stay away from dermal contact with cyanide-sullied patients or with gastric substance of patients who might have ingested cyanide-containing materials, Patients don’t present auxiliary pollution chances later tainted dress is eliminated and the skin is washed.
Hydrogen cyanide harming is set apart by sudden beginning of significant poisonous impacts that might incorporate syncope, seizures, extreme lethargies, panting breaths, and cardiovascular breakdown, causing demise in practically no time.
Patients presented to hydrogen cyanide can make due with strong consideration and fast organization of explicit cures.
Cleaning Region
Recently cleaned patients and patients presented uniquely to hydrogen cyanide gas who have no skin or eye bothering might be moved promptly to the Basic Consideration Region. Different patients require purification as depicted underneath.
ED staff should wear butyl elastic gloves and covers prior to treating patients who have been presented to hydrogen cyanide fluid or arrangements. (Hydrogen cyanide promptly enters most rubbers and obstruction textures or creams, yet butyl elastic gives great skin insurance to a brief timeframe.)
Know that utilization of defensive hardware by the supplier might cause dread in youngsters, bringing about diminished consistence with additional administration endeavors.
As a result of their generally bigger surface area:body weight proportion, kids are more helpless against poisons ingested through the skin. Additionally, trauma center staff ought to look at kids’ mouths due to the recurrence of hand-to-mouth movement among youngsters.
ABC Updates
Assess and support aviation route, breathing, and flow. In instances of respiratory trade off secure aviation route and breath through endotracheal intubation. If impractical, carefully make an aviation route.
Patients who are torpid, hypotensive, or have seizures or heart dysrhythmias ought to be treated in the regular way. If not recently directed, give sodium bicarbonate intravenously to these patients. Further bicarbonate treatment ought to be directed by ABG estimations.
Essential Cleaning
Patients who are capable may help with their own purification.
Speed is basic. On the off chance that the patient is suggestive, promptly foundation crisis life support measures, including the utilization of the cyanide cure pack (see Remedys and Different Medicines underneath).
Assuming that the patient’s clothing is wet with hydrogen cyanide arrangement, as soon as possible take off tainted dress while flushing uncovered skin and hair with plain water for 2 to 3 minutes (ideally under a shower), then, at that point, wash twice with gentle cleanser.
Use alert to stay away from hypothermia while purifying youngsters or the older. Use covers or warmers when suitable.
Flush completely with water. Twofold pack tainted dress and individual effects.
Water uncovered eyes for something like 5 minutes. Eliminate contact focal points if effectively removable without extra injury to the eye. Proceed with water system while shipping the patient to the Basic Consideration Region.
In instances of ingestion, don’t actuate emesis. Assuming actuated charcoal has not been directed beforehand, and the casualty is ready, asymptomatic, and has a gag reflex, regulate a slurry of enacted charcoal (oversee at 1 gm/kg, normal grown-up portion 60-90 g, youngster portion 25-50 g).
A soft drink can and a straw might be of help when offering charcoal to a youngster. Think about gastric lavage on the off chance that the patient is cognizant and it very well may be performed not long after ingestion.
Since cyanide retention from the stomach is fast, the viability of actuated charcoal will rely upon how rapidly later ingestion it tends to be directed. Disconnect gastric washings and vomitus; they might off-gas hydrogen cyanide.
Basic Consideration Region
Be sure that purification has been done (see Disinfecting Region above).
ABC Updates
Assess and support aviation route, breathing, and dissemination as in ABC Updates above. Set up intravenous access in genuinely sick patients in the event that this has not been done beforehand. Persistently screen cardiovascular mood.
Patients who are in stun or have seizures ought to be treated by ALS conventions. These patients or the individuals who have dysrhythmias might be genuinely acidotic; think about giving 1 mEq/kg intravenous sodium bicarbonate.
Inward breath Openness
Inward breath is the essential course of openness to hydrogen cyanide. Allude to Cures and Different Medicines beneath for clinical treatment of foundational impacts.
Skin Openness
Assuming the skin reached hydrogen cyanide fluid or cyanide arrangements, synthetic consumes may happen; treat as warm consumes. Watch for signs or indications of foundational harmfulness, which might be postponed in beginning for as long as 60 minutes.
Eye Exposure
Proceed with water system for something like 15 minutes. Test visual sharpness. Look at the eyes for corneal harm and treat suitably. Promptly counsel an ophthamologist for patients who have corneal wounds.
Ingestion Exposure
Try not to actuate emesis. Assuming the casualty is indicative, promptly establishment crisis life support measures including the utilization of a cyanide remedy pack (see Antitoxins and Different Medicines underneath).
On the off chance that the casualty is ready, asymptomatic, has a gag reflex, and it has not been done already, perform gastric lavage and give actuated charcoal quickly. Since cyanide retention from the stomach is fast, the value of actuated charcoal will rely upon how rapidly later ingestion it tends to be managed.
Direct a slurry of initiated charcoal at 1 gm/kg (regular grown-up portion 60-90 g, kid portion 25-50 g). A soft drink can and a straw might be of help when offering charcoal to a kid.
Harmful vomitus or gastric washings ought to be segregated (e.g., by appending the lavage cylinder to separated divider pull or one more shut compartment).
Antitoxins and Different Medicines
Patients who have signs or indications of huge fundamental harmfulness ought to be assessed for curing treatment.
In the US, cures for cyanide incorporate amyl nitrite perles and intravenous imbuements of sodium nitrite and sodium thiosulfate, which are bundled in the cyanide remedy unit.
How many electron groups are there in HCN?
Valence Shell Electron Pair Repulsion is:
Number of Electron Groups Electron Group Geometry Example 2 Linear HCN 3 Trigonal planar BF3 3 Trigonal planar SO2
What is the full name of H3PO4?
Orthophosphoric acid
Orthophosphoric acid , H3PO4, is usually called simply phosphoric acid.
Sumamry
Assuming that one portion of the cures from the pack has been regulated already by prehospital faculty and deficient clinical reaction has happened, a second portion of one-a large portion of the underlying sums might be given 30 minutes later.
Frequestly Ask Questions
Here, some question described related to this article.
### 1. What is hydrogen cyanide?
At room temperature, hydrogen cyanide is an unpredictable, lackluster to-blue fluid (additionally called hydrocyanic corrosive). It quickly turns into a gas that can create demise in minutes whenever relaxed. Hydrogen cyanide is utilized in making filaments, plastics, colors, pesticides, and different synthetic substances, and as a fumigant to kill rodents. It is likewise utilized in electroplating metals and in creating visual film.
2. What prompt wellbeing impacts can be brought about by openness to hydrogen cyanide?
Breathing limited quantities of hydrogen cyanide might cause cerebral pain, dazedness, shortcoming, sickness, and regurgitating. Bigger sums might cause wheezing, unpredictable pulses, seizures, blacking out, and surprisingly fast passing. By and large, the more genuine the openness, the more extreme the indications. Comparative manifestations might be created when arrangements of hydrogen cyanide are ingested or interacted with the skin.
3.Will hydrogen cyanide harming be dealt with?
The treatment for cyanide harming incorporates breathing unadulterated oxygen, and on account of genuine manifestations, treatment with explicit cyanide antitoxins. People with genuine manifestations should be hospitalized.
4.Are any future wellbeing impacts liable to happen?
A solitary little openness from which an individual recuperates rapidly isn’t probably going to cause deferred or long haul impacts. Later a genuine openness, a patient might have mind or heart harm.
5.What tests should be possible assuming an individual has been presented to hydrogen cyanide?
Explicit tests for the presence of large are not valuable to the specialist. On the off chance that a serious openness has happened, blood and examinations and different tests might show whether the mind or heart has been harmed. Testing isn’t required for each situation.
6.Where can more data about hydrogen cyanide be found?
More data about hydrogen cyanide can be acquired from your provincial toxin control focus; your state, district, or nearby wellbeing division; the Office for Poisonous Substances and Illness Library (ATSDR); or a center in your space that represents considerable authority in word related and ecological wellbeing.
Assuming that the openness occurred working, you might wish to examine it with your manager, the Word related Security and Wellbeing Organization (OSHA), or the Public Foundation for Word related Wellbeing and Wellbeing (NIOSH). Ask the individual who gave you this structure for help in finding these phone numbers.
7.For what reason is HCN a feeble corrosive?
Solid acids have feeble form bases. The way that HCN is a solid corrosive infers that its form base Cl–is too feeble a base to clutch the proton in rivalry with one or the other H2O or H3O+. Additionally, the CN–particle ties unequivocally to a proton, making HCN a powerless corrosive
8.Is HCN polar?
Yes, HCN is a polar particle on the grounds that there is a huge electronegative distinction between the N and H across the straight atom. It comprises of two polar bonds whose polarities line up a similar way.
9. What’s the name for HF?
Hydrogen fluoride is a substance compound that contains fluorine.
10. Is HCN a solid electrolyte?
HCN is delegated a feeble electrolyte. This is on the grounds that when disintegrated in water, a low level of the HCN atoms ionize. HCN isn’t one of the solid acids and subsequently would be one of the feeble acids (which are powerless electrolytes). Solid electrolytes would require total ionization/separation.
11. Is HCN a preferable corrosive over water?
Of these acids the most grounded is HF as it has the biggest Ka. Hydrocyanic is the most fragile of the gathering as it has the littlest Ka. This implies that HF will separate indeed when set in water than different acids. HCN will separate the least.
-
Corrosive (Base) Strength.
-
Corrosive Name
-
Equation
-
Ka
-
Hydrocyanic
-
HCN
-
4.9 x 10-10
12. What sort of bond is HCN?
In HCN, Carbon is clung to Nitrogen with a triple covalent bond comprising of one sigma bond and two pi bonds. The sigma bond is shaped by covering hybridized orbitals, with the two leftover unhybridized orbitals covering to frame the two pi bonds.
13.What is the name of h_3po_4?
The name of the compound H3PO4 is phosphoric corrosive. This synthetic compound is comprised of three hydrogen iotas, one phosphorous particle and four oxygen
14. Who found hydrogen fluoride?
French scientific expert Edmond Frémy found HF gas during the nineteenth century, in spite of the fact that HF arrangements had been known and utilized something like 100 years sooner. Today principle source is the mineral fluorite (calcium fluoride), which is treated with sulfuric corrosive to shape HF gas.
15. Is CH3CH2OH a solid electrolyte?
No, Ethanol, CH3CH2OH, is a nonelectrolyte; smelling salts, NH3, is a powerless electrolyte; and scandium(III) sulfate, Sc2(SO4)3, is a solid electrolyte.
Conclusion
If anyone wants to about HCN. Here, I describe that HCN is the name of Hydrogen cyanide. In this article, I fully describe related to Hydrogen cyanide (HCN). Hopefully this article will helpful for you .