Sodium Sulfate Formula Is Na2SO4. The sodium salt of sulfuric acid is sodium sulfate (Na2SO4). It is an odorless crystalline white solid. Its molecular weight in anhydrous form is142.04gm/mole. It is soluble in water.

What Is Sodium Sulfate, And What Does It Do?
The sodium salt of sulfuric acid is sodium sulfate (Na2SO4). Anhydrous sulfate, also known as thenardite, is a white crystalline solid, whereas the decahydrate Na2SO4.10H2O is known as Glauber’s salt or mirabilis.
When Na2SO4.7H2O is cooled, it transforms into mirabilite. The natural mineral form of the decahydrate is mirabilite. Mirabilite accounts for almost two-thirds of global sodium sulfate output. It’s also made from by-products of chemical processes like the manufacturing of hydrochloric acid.
Anhydrous Means “Without Water.”
The term “anhydrous” literally means “without water.” In chemistry, compounds that are devoid of water are referred to as anhydrous. The word is most commonly used to describe crystalline substances after the crystallization water has been removed.
The presence or absence of water in the crystalline structure of salt compounds changes the name and function of the compound. When sodium sulfate, NaSO4, is devoid of water, it is known as anhydrous sodium sulfate and is utilized as a drying medium.
The decahydrate form of the same substance is known as “Glauber’s salt,” and it’s used to create glass. Salt absorbs moisture when it comes into touch with it since it does not have water bound to its molecules, which is why it is used as a drying agent.
Table Of Contents
| 1. Formula | Na2SO4 |
|---|---|
| 2. Molar mass | 142.04 g/mol |
| 3. Soluble in | Water |
| 4. Boiling point | 1,429 °C (2,604 °F; 1,702 K) (anhydrous) |
| 5. Appearance | white crystalline solid; hygroscopic |
| 6. Density | 2.664 g/cm3 (anhydrous); 1.464 g/cm3 (decahydrate) |
| 7. Melting point | 884 °C (1,623 °F; 1,157 K) (anhydrous); 32.38 °C (decahydrate) |
Sodium Sulfate’s Characteristics
- The formula for a molecule Na2SO4
Molecular weight (anhydrous):
- 142.04gm/mole, 322.20gm/mole (decahydrate)
Appearance
- Crystalline white solid
Odor
-
Odorless
Boiling point -
1429oC is the boiling point (anhydrous)
-
Flashpoint 800oC
-
884°C melting point (anhydrous), 32.40°C melting point (decahydrate)
-
1.464gm/ml (anhydrous), 2.664gm/ml (anhydrous) (decahydrate)
Refractive index:
-
1.468 (anhydrous)
-
1.394 (non-anhydrous) (decahydrate)
Solubility
-
Glycerol and glycerol esters are soluble in water.
-
Iodide of hydrogen and insoluble in water
Preparation Of Sodium Sulfate
Johann Rudolf Glauber found sodium sulfate in spring water in Austria in 1625, and the hydrate form is known as Glauber’s salt. He termed it sal mirabilis because of its therapeutic virtues (miraculous salt).
The chemical industry produces one-third of the world’s sodium sulfate as a by-product of other processes. The interaction between sodium chloride with sulfuric acid creates it.
- 2 HCl + Na2SO4 2 NaCl + H2SO4
Glauber’s salt
Until the 1900s, the crystals were employed as a general-purpose laxative. Glauber’s salt was utilized as a raw material for the industrial manufacturing of soda ash in the 18th century by reacting with potassium carbonate or potash.
As the demand for soda ash grew in the nineteenth century, the large-scale Leblanc process, which created synthetic sodium sulfate, became the primary technique of soda ash manufacturing.
Excretion is primarily in the urine at dietary amounts. Sulfates are found in all body cells, with connective tissues, bone, and cartilage having the largest quantities. Sulfates are engaged in several essential metabolic pathways, including those that are involved in detoxification.
-
Natural sodium sulfate and by-product sodium sulfate, often known as synthetic sodium sulfate, are the two forms.
-
Natural sodium sulfate is generated in California and Texas from naturally occurring brines and crystalline deposits.
-
Saline lakes, such as Utah’s Great Salt Lake, have it as a constituent. As a byproduct of many manufacturing operations, synthetic sodium sulfate is recovered.
-
Both forms of sodium sulfate have a variety of essential and practical uses in consumer goods.
-
Sodium sulfate was placed 47th in terms of quantity produced in a survey of the top 50 basic organic and inorganic compounds produced in the United States.
Sodium Sulfate Resources
Sodium is the most abundant element in the Earth’s crust comes on number 6. Mineral formations containing sodium sulfate are geologically young, mostly post-glacial.
-
Sodium sulfate is a common component of seawater and many saline or alkaline lakes and is found in abundance.
-
Natural sodium sulfate reserves are estimated to be 3.3 billion tons globally.
-
Natural sodium sulfate reserves are sufficient to supply predicted demand for several millennia, with global output averaging around 2.6 million tons per year.
-
The amount of synthetic sodium sulfate produced is determined by the manufacturing businesses’ ability to recover product sulfate over time.
-
Surface depressions or lakes with no exits that are fed by spring waters running over volcanic rocks containing sulfide minerals frequently produce soluble sulfide salts that are oxidized by air to produce sulfates.
Sodium Sulfate’s Applications
1. Organic liquids are dried with sodium sulfate.
2. In-home laundry detergents as a filler.
3. As a fining agent for molten glass, it removes microscopic air bubbles.
4. Glauber’s salt, decahydrate, was employed as a laxative to eliminate certain medicines from the body, such as acetaminophen.
5. Defrosting windows, carpet fresheners, starch production, and animal feed addition
6. In the production of detergents and the Kraft paper pulping process.

What Happens When Sodium Sulfate Reacts With Barium Chloride?
In a double displacement reaction, sodium sulfate interacts with barium chloride to produce barium sulfate and sodium chloride.
BaCl2 + Na2SO4 = BaSO4 + 2NaCl = BaSO4 + 2NaCl = BaSO4 + 2NaCl = BaSO4 + 2Na
Because sodium sulfate is an electrostatically coupled ionic sulfate, this reaction happens.
Summary
The sodium salt of sulfuric acid is sodium sulfate (Na2SO4). NaSO4 is devoid of water and is used as a drying agent. The decahydrate form of the same substance is known as “Glauber’s salt,” and it’s used to create glass.
Molecular Weight (anhydrous): 142.04gm/mole, 322.20gaule (decahydrate), 2.664gm/ml (a). Glycerol and glycerol esters are soluble in water; sodium sulfate can be evaporated.
Write A Brief Remark About Sodium Sulfate’s Water Solubility.
At 0 degrees Celsius, anhydrous sodium sulfate dissolves in water at a rate of 47.6 grams per liter. Its solubility skyrockets to 139 grams per liter when heated to 20 degrees Celsius. Finally, the solubility of sodium sulfate in water at a temperature of 100 degrees Celsius is 427 grams per liter.
What Is The Best Way To Make Sodium Sulfate?
1. The Mannheim procedure can be used to make sodium sulfate, which involves the following reaction:
- Na2SO4 + 2HCl = H2SO4 + 2NaCl
2. It can alternatively be made using the Hargreaves method, which is outlined below:
2Na2SO4 + 4HCl = 4NaCl + 2H2O + 2SO2 + O2
Forms Of Sodium Sulfate
There are 3 forms of sodium sulfate:
1. The rare mineral thenardite is anhydrous sodium sulfate, which is utilized as a drying agent in chemical synthesis.
2. Heptahydrate sodium sulfate is an extremely unusual type of sodium sulfate.
3. The mineral mirabilite, which is decahydrate sodium sulfate, is frequently employed in the chemical industry. Glauber’s salt is another name for it.
History Of Sodium Sulfate
Glauber’s salt is a decahydrate of sodium sulfate named after Johann Rudolf Glauber (1604–1670), a Dutch/German chemist and apothecary who discovered it in Austrian spring water in 1625.
Because of its medical powers, he termed it sal mirabilis (miracle salt):
- The crystals were employed as a general-purpose laxative until more sophisticated alternatives emerged in the 1900s.
Glauber’s salt was first employed as a raw material for the commercial manufacturing of soda ash (sodium carbonate) in the 18th century when it was combined with potash (potassium carbonate). Soda ash demand increased, and sodium sulfate supply had to grow in response.
As a result, the large-scale Leblanc process, which produces synthetic sodium sulfate as a critical step, became the primary method of soda-ash production in the nineteenth century.
Chemical Characteristics Of Sodium Sulfate
1. A typical electrostatically bound ionic sulfate is sodium sulfate. The easy production of insoluble sulfates when these solutions are treated with Ba2+ or Pb2+ salts indicates the presence of free sulfate ions in the solution:
- 2 NaCl + BaSO4 + Na2SO4 + BaCl2
2. Most oxidizing and reducing agents do not react with sodium sulfate. It can be transformed to sodium sulfide by carbothermal reduction (also known as thermo-chemical sulfate reduction (TSR), high-temperature heating with charcoal, and so on) at high temperatures:
- 2 CO2 + 2 SO4 + 2 SO4 + 2 SO4 + 2 SO4 + 2 SO4 + 2 SO4 + 2 SO4 + 2 SO4 + 2 SO4
The Leblanc process, a now-defunct industrial route to sodium carbonate, used this reaction.
3. The acid salt sodium bisulfate is formed when sodium sulfate interacts with sulfuric acid:
- 2 NaHSO4 = Na2SO4 + H2SO4
Sodium Sulfate Has A Moderate Proclivity For Forming Double Salts.
In contrast to potassium sulfate and ammonium sulfate, which generate many stable alums, the only alums formed with common trivalent metals are Naal(SO4)2 (unstable beyond 39 °C) and NaCr(SO4)2. Some other alkali metal sulfates, such as Na2SO43K2SO4, which occurs naturally as the mineral aphthitalite, have double salts.
The interaction of sodium sulfate with potassium chloride to form glaserite has been utilized to develop a method for manufacturing potassium sulfate, a fertilizer.
1. 3Na2SO4CaSO4
2. 3Na2SO4MgSO4 (vanthoffite)
3. NaFNa2SO4 are some of the other double salts.
Physical Characteristics
In water, sodium sulfate exhibits remarkable solubility properties.
-
Between 0 and 32.384 °C, its solubility in water increases by more than tenfold, reaching a high of 49.7 g/100 ml.
-
The slope of the solubility curve changes at this point, and the solubility becomes almost temperature independent.
-
This temperature of 32.384 °C, which corresponds to the release of crystal water and the melting of the hydrated salt, is used to calibrate thermometers.
-
Solubility of Na2SO4 in water following as a function of temperature
Structure Of Sodium Sulfate
The decahydrate crystals are made up of [Na(OH2)6]+ ions with octahedral molecular geometry. Eight of the ten water molecules are coupled to sodium in these octahedra, while two others are interstitial, hydrogen-bonded to sulfate. Hydrogen bonds connect these cations to the sulfate anions.
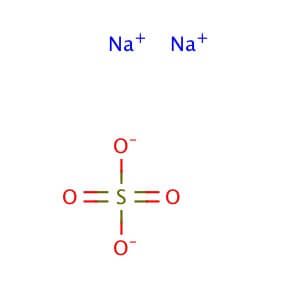
The distances between Na and O are around 240 pm. The residual entropy (entropy at absolute zero) of crystalline sodium sulfate decahydrate is 6.32 kJ/(mol), which is exceptional among hydrated salts. This is due to its ability to transfer water faster than most other hydrates.
Production Of Sodium Sulfate
The global output of sodium sulfate, nearly entirely in the form of decahydrate, is estimated to be between 5.5 and 6 million tons per year (Mt/a). In 1985, production was 4.5 Mt/a, with half of it coming from natural sources and the other half from chemical sources.
Natural production climbed to 4 Mt/a after 2000, remained consistent until 2006, and chemical production declined to 1.5 to 2 Mt/a, for a total of 5.5 to 6 Mt/a. Natural sodium sulfate and sodium sulfate manufactured chemically are nearly identical in all uses.
Summary
Sodium sulfate can be made using the Mannheim or Hargreaves methods. The mineral mirabilite, which is decahydrate sodium sulfate, is another name for it. Sodium sulfate has a moderate proclivity for forming double salts.
Between 0 and 32.384 °C, its solubility in water increases by more than tenfold. The interaction of sodium sulfate with potassium chloride to form glaserite has been utilized to develop a fertilizer.
Sources Derived From Nature
The natural mineral form mirabilite, which can be found in lake beds in southern Saskatchewan, produces two-thirds of the world’s decahydrate (Glauber’s salt). Mexico and Spain were the world’s largest producers of natural sodium sulfate in 1990 (each approximately 500,000 tons), followed by Russia, the United States, and Canada (each around 350,000 tons).
Over a billion tons of natural resources are believed to be available. Searles Valley Minerals (California, US), Airborne Industrial Minerals (Saskatchewan, Canada), Qumica del Rey (Coahuila, Mexico).
Minera de Santa Marta (Toledo, Spain), Sulquisa (Madrid, Spain), Chengdu Sanlian Tianquan Chemical (Tianquan County, Sichuan, China), Hongze Yinzhu Chemical Group (Hongze, China) (Altai Krai, Siberia, Russia).
The mineral thenardite is formed from anhydrous sodium sulfate in arid settings. In wet air, it slowly transforms into mirabilite. Believerite, a calcium sodium sulfate mineral, is another form of sodium sulfate. Neither mineral is as common as mirabilite.
Industry Of Chemicals
Sodium sulfate is produced as a by-product of other chemical processes for around a third of the world’s sodium sulfate. The majority of this output is chemically inherent in the main process and is only moderately cost-effective. As a result of the industry’s efforts, the output of sodium sulfate as a by-product is decreasing.
The most important chemical source of sodium sulfate is hydrochloric acid, which can be made from sodium chloride (salt) and sulfuric acid in the Mannheim process or sulfur dioxide in the Hargreaves process.
1. The salt cake is the sodium sulfate that results from these operations.
-
2 HCl + Na2SO4 = 2 NaCl + H2SO4 = 2 HCl + Na2SO4 = 2 HCl + Na2SO4 = 2 HCl + Na2SO4 = 2 HC
-
4 HCl + 2 Na2SO4 = 4 HCl + 2 Na2SO4 = 4 HCl + 2 Na2SO4 = 4 HCl + 2 Na2SO4 = 4 HCl + 2 Na2SO4 = 4 HCl + 2 Na2SO4 = 4 HCl +
2. The second significant source of sodium sulfate is the process of neutralizing excess sodium hydroxide with sulfuric acid, which is used extensively in the rayon industry. This procedure is also a commonly used and practical laboratory preparation.
- 2 NaOH(aq) + H2SO4(aq) + 2 H2O(l) H = -112.5 kJ Na2SO4(aq) + 2 H2O(l) (highly exothermic)
3. It can also be made in the lab by reacting sodium bicarbonate with magnesium sulfate.
- Na2SO4 + Mg(OH)2 + 2 CO2 = 2 NaHCO3 + MgSO4 = Na2SO4 + Mg(OH)2 = Na2SO4 + Mg(OH)2 = Na2SO4 + Mg(OH)
Laboratory Synthesis Of Sodium Sulfate
Laboratory synthesis, on the other hand, is not commonly used because commercial supplies are readily available. Sodium sulfate was previously a by-product of the production of sodium dichromate, which involves adding sulfuric acid to a sodium chromate solution to produce sodium dichromate or chromic acid.
In the synthesis of lithium carbonate, chelating agents, resorcinol, ascorbic acid, silica pigments, nitric acid, and phenol, sodium sulfate is or was generated. The decahydrate form of sodium sulfate is commonly used to purify bulk sodium sulfate since the anhydrous form attracts iron compounds and organic molecules. By gently warming the hydrated form, the anhydrous form can be easily created.
In 2006, Elementis Chromium (chromium industry, Castle Hayne, NC, US), Lenzing AG (200 Mt/a, rayon industry, Lenzing, Austria), Addison (formerly Rhodia, methionine industry, Les Roches-Roussillon, France), Elementis (chromium industry, Stockton-on-Tees, UK), Shikoku Chemicals (Tokushima, Japan), Visko-R (Visko (rayon industry, Russia).
Applications Of Sodium Sulfate
Sodium sulfate is a drying agent for organic liquids. Clumps occur here, suggesting that water is present in the organic liquid. The liquid can be brought to dryness by adding more sodium sulfate, as shown here by the lack of clumping.
Industries That Deal With Commodities
Sodium sulfate is a very inexpensive commodity, with US prices as low as $30 per ton in 1970, rising to $90 for salt cake quality and $130 for better grades. The most common application is as a filler in powdered home laundry detergents, which accounts for around half of global output.
Domestic consumers are rapidly converting to compact or liquid detergents that do not include sodium sulfate, which is reducing their use. Another once-common usage of sodium sulfate, particularly in the United States and Canada, is in the Kraft process for producing wood pulp.
The organics in the “black lliquid” produced by this process are burned to generate heat, which is required to drive the conversion of sodium sulfate to sodium sulfide. However, in the early 1960s, improvements in the thermal efficiency of the Kraft recovery process allowed for more efficient sulfur recovery, reducing the requirement for sodium sulfate makeup.
As a result, the use of sodium sulfate in the pulp sector in the United States and Canada has decreased from 1,400,000 tons per year in 1970 to around 150,000 tons in 2006. Another important application for sodium sulfate is the glass sector, which is Europe’s second-largest.
Fining agents, such as sodium sulfate, are used to eliminate microscopic air bubbles from molten glass. It fluxes the glass and keeps the glass melt from forming scum during refining. From 1970 to 2006, Europe’s glass sector consumed a consistent 110,000 tons each year.
Summary
Mexico and Spain were the world’s largest producers of natural sodium sulfate in 1990. Russia, the United States, and Canada were each around 350,000 tons. Over a billion tons of natural resources are believed to be available for industrial production.
The majority of this output is chemically inherent in the main process and is cost-effective. Sodium sulfate was previously a by-product of the production of sodium dichromate.
Sodium Sulfate Is Used In The Production Of Textiles
Sodium sulfate is used in the production of textiles, especially in Japan, where it is the most common application. Sodium sulfate is added to improve the ionic strength of the solution, which aids in “leveling,” or the reduction of negative electrical charges on textile fibers, allowing dyes to absorb uniformly.
It does not damage the stainless steel vessels used in dyeing, unlike the alternative sodium chloride. In 2006, this use consumed around 100,000 tons in Japan and the United States.
Industry Of Food
Sodium sulfate is used in following:
1. The diluent sodium sulfate is used to dilute food colors.
2. It’s referred to as E number additive E514.
Storage Of Heat
This material is particularly well suited for storing low-grade solar heat for subsequent release in space heating applications because of its high heat storage capacity during the phase transition from solid to liquid and the favorable phase change temperature of 32 °C (90 °F).
In some cases, the salt is mixed into thermal tiles that are installed in attics, while in others, the salt is put into cells that are surrounded by solar-heated water. The phase change provides for a significant reduction in the quantity of material needed for effective heat storage, as well as temperature consistency as long as enough material in the right phase is available.
A mixture with common sodium chloride salt (NaCl) lowers the melting point to 18 °C (64 °F) for cooling applications. The fusion heat of NaClNa2SO410H2O has been marginally raised to 286 kJ/kg.
Summary
Sodium sulfate is used in the production of textiles, especially in Japan. It is particularly well suited for storing low-grade solar heat for subsequent release in space heating applications. In 2006, this use consumed around 100,000 tons in Japan and the U.S.
Applications Of Sodium Sulfate On A Small Scale
Anhydrous sodium sulfate is a common inert drying agent in the laboratory for eliminating traces of water from organic solutions.
-
It is more effective than magnesium sulfate, although it takes longer to work. It is only effective below about 30 °C, but because it is chemically rather inert, it can be utilized with a range of materials. Sodium sulfate is added to the solution until the crystals no longer clump together.
-
The decahydrate of Glauber’s salt is used as a laxative. It is efficient in removing certain medicines from the body, such as paracetamol (acetaminophen), such as after an overdose.
-
Sodium sulfate was proposed in 1953 as a heat storage material for passive solar heating systems. Its remarkable solubility qualities, as well as the high heat of crystallization (78.2 kJ/mol), are used to achieve this.
-
Other applications for sodium sulfate include defrosting windows, starch production, carpet freshener additives, and livestock feed additives.
-
Thermaltake, for example, produces a laptop computer chill mat (soft Notebook Cooler) that contains sodium sulfate decahydrate in a quilted plastic pad. The substance slowly degrades into a liquid and circulates, balancing the temperature of the laptop and acting as an insulator.
Safety Required For Sodium Sulfate
Sodium sulfate is generally considered to be non-toxic, however, it should be handled with caution. Dust can cause brief asthma or eye irritation, which can be avoided by using eye protection and wearing a paper mask. There are no restrictions on transportation, and no Risk or Safety Phrases apply.
Sodium sulfite isn’t used to clean anything, at least not in the laundry. It’s added to prevent corrosion in the washing machine or dishwasher. Rust is formed when iron combines with oxygen to generate ferric oxide. High temperatures, such as those found in washing machines, facilitate this process.
What Is The Source Of Oxygen?
It dissipates in water. Because water’s surface is in contact with air, oxygen can always dissolve. Photosynthesis, which occurs while aquatic plants grow, also produces oxygen as a byproduct.
The amount of oxygen dissolved is affected by temperature (fewer dissolves as temperature rises), pressure (fewer dissolves at higher altitudes), and the number of other chemicals already dissolved in the water (freshwater holds more oxygen than saltwater).
Water with oxygen is beneficial for fish, who get their oxygen by extracting the gas when water passes through their gills, but it’s bad for metals in washing machines, which corrode. The sulfite of sodium is an oxygen scavenger.
It forms sodium sulfate when it combines with oxygen, thereby lowering the dissolved oxygen content and preserving the insides of washing machines from rusting. Small quantities can be disposed of by flushing them down the drain.
If just minute amounts of the following substances are present, they can be flushed down the drain:
-
Light metal salts, such as calcium chloride or sodium sulfate
-
Acids made from minerals (Hydrochloric acid, sulfuric acid, phosphoric acid)
-
Sodium hydroxide and potassium hydroxide are two different types of hydroxide.
What Does It Mean To Have A “Little Quantity”?
This is a question that no one will be able to answer. Everyone understands that modest numbers cause fewer environmental issues. However, because no one can calculate the environmental issues, you will almost always be told, “In case of doubt, discard as toxic waste.”
The lower the tolerable quantities, the more stupid your counsel is because every council is afraid of giving incorrect advice, which could result in a criminal deed in the end. You’ll find yourself in unusual circumstances, possibly even frantic, and you’ll have to make your own choices.
Summary
Sodium sulfate is considered non-toxic but should be handled with caution. It can be used as a heat storage material for passive solar heating systems and as a laxative. The decahydrate of Glauber’s salt is used as an effective drying agent in the laboratory. Water with oxygen is beneficial for fish but bad for metals in washing machines.
What Is The Environmental Impact Of Sodium Sulfate?
When it comes to mercury, you’ll fight tooth and nail to keep every milligram from escaping into the wastewater. You can be much more generous with sodium sulfate. If you dispose of toxic waste, think about the consequences.
For example, when sodium sulfate is burned at 1200 degrees Celsius, sulfur trioxide is produced, which escapes with the exhaust stream and must be filtered out. OK, you can make gypsum boards with the filtered material, but the slag must be disposed of underground.
Consider whether the salting of the water is a small issue when compared to disposal of sodium sulfate with wastewater.
Maximum concentrations for numerous compounds have been established by local legislation, mainly for heavy metal salts such as mercury, but also for copper. Because you don’t know the concentration of wastewater from the entire institute building when it’s dumped into the sewerage water system, the particular values won’t help you.
If your “little quantity” isn’t a problem, it could assist to know that it’s sometimes referred to as “small quantity typical for usage in a lab.” This refers to quantities of less than one kilogram.
Is Sodium Sulfate A Skin Safe Ingredient?
In terms of its use in cosmetics and body goods, the SLS safety assessment study (the most recent assessment) was published in 1983 in the International Journal of Toxicology.
It was discovered that if applied briefly and rinsed off the skin, as with shampoos and soaps, it is not hazardous.
Is Sodium Benzoate Poisonous?
While sodium benzoate is deemed harmless, scientists have discovered that when it is combined with ascorbic acid, it has detrimental side effects (vitamin C). According to their research, it subsequently converts to benzene, a recognized carcinogen that can cause cancer.
Is it safe to eat sodium sulfite?
Sulfites are a preservative to which many people are allergic and which can worsen asthma. Sulfites are not allowed to be used on fresh fruits and vegetables in the United States, although they can be found in other foods.
Frequently Asked Questions
People usually ask following questions.
1, In sodium sulfate, which acid is present?
Sulfuric acid is an acid that contains sulfur. The sodium salt of sulfuric acid is known as sodium sulfate. The chemical formula for sodium sulfate is Na2SO4.
2. What are the risks associated with sodium sulfate?
It’s not flammable. In a fire, it emits annoying or hazardous fumes (or gases). Use appropriate extinguishing media if there is a fire in the vicinity.
3. What is the molar mass of sodium sulfate (Na2SO4)?
142.04 g/mol is the molecular weight of 142.04 g/mol
The molar mass of sodium sulfate
The formula Na2SO4 states that one mole of sodium sulfate is made up of two mole’s of sodium (45.98 g), one-mole sulfur (32.06 g), and four mole’s of oxygen (64.00 g) (142.04 g).
4. Is Na2SO4 a polar or a nonpolar substance?
The sulfate anion is generated by covalent interaction between sulfur and oxygen, and the formula for this chemical is [Na+]2[SO42-]. S-O bonds are polar covalent bonds because they have a bond polarity.
5. Is it safe for you to handle sodium sulfate?
Avoid coming into touch with your eyes, skin, or clothing. Storage: Keep in a cool, dry location. Protect from moisture when storing. PELs that have been vacated by OSHA: Sulfate of sodium: There are no OSHA Vacated PELs for this compound.
6. Is NaCl a polar salt?
An ionic compound, sodium chloride (NaCl), acts as a polar molecule. The considerable disparity in electronegativities between sodium and chlorine typically causes their bond to be polar. Furthermore, in sodium chloride, Na has a +1 charge while Cl has a -1 charge, forming a strong connection.
7. Why does sodium chloride dissolve in water?
When salt is mixed with water, the covalent bonds of water are stronger than the ionic bonds in the salt molecules, and the salt dissolves. The ionic link that held the sodium and chloride ions together is broken as water molecules pull them apart.
8. Where can you find sulfate?
Sulfate is a naturally occurring chemical. It can be found in varying levels of water. The water may have a harsh taste if it has a high degree of sulfate. Minerals, soil, rocks, plants, and food all contain sulfates.
9. What is the meaning of the word sulfate?
Sulfate is a chemical with four oxygen atoms surrounding a sulfur atom. With bonding, the sulfate molecule forms a tetrahedral structure. When sulfate is turned to sulfuric acid, it generates salt.
10. Who was the first to find sulfate?
The discovery of sodium sulfate by Johann Glauber - Sal Mirabile Glauberi | Journal of Chemical Education.
11. Why does salt cause ice to melt?
The fact that a solution of water plus dissolved salt has a lower freezing point than pure water is the real reason why salt causes ice to melt. As a result, ice that comes into touch with salty water melts, resulting in more liquid water, which dissolves more salt, causing more ice to melt, and so on.
12. What is the purpose of sodium sulfate?
Sodium sulfate, often known as Na2SO4, is a white crystalline solid or powder used in the production of craft paper, paperboard, glass, and detergents, as well as a raw ingredient for numerous chemicals.
13. Is sodium sulfate dangerous?
The acute toxicity of sodium sulfate (LD50) has not been determined with certainty, however, it is most likely more than 5000 mg/kg. At 10 mg/m3, no adverse effects were seen in an aerosol inhalation study. Human data also show that sodium sulfate has a very low acute toxicity.
14. What is sodium sulfate and where does it come from in food?
The diluent sodium sulfate is used to dilute food colors. It’s referred to as E number additive E514.
15. Is sodium sulfate beneficial to the skin?
It turns out that sulfates can be overly successful in removing oil, leaving hair and skin dried. Sulfates can also cause irritations like redness and itching if your skin or scalp is very sensitive.
17. Why are sulfates harmful to your health?
Sulfates are a major component of haze pollution, which irritates the lungs and has an impact on lung health. Skin that is parched. Sulfates in consumer products are known to cause dry skin. Sulfates can be found in laundry detergent, soap, and shampoo.
18. Is sodium sulfate safe to consume?
The dry powder should not be swallowed. Before using, give it a good shake. Unless your doctor tells you otherwise, drink plenty of noncaffeine liquids before, during, and after using sodium sulfate, potassium sulfate, or magnesium sulfate.
19. Is there a distinction between sulfites and sulfates?
Sulfates and sulfites are both sulfur-containing chemicals. Sulfates are sulfuric acid salts that you undoubtedly come across regularly. Sulfites are naturally occurring chemicals found in all wines that work as preservatives by limiting the growth of microorganisms.
Conclusion
The sodium salt of sulfuric acid is sodium sulfate (Na2SO4). NaSO4 is devoid of water and is used as a drying agent. The decahydrate form of the same substance is known as “Glauber’s salt,” and it’s used to create glass.
Sodium is the most abundant element in the Earth’s crust comes on number 6. Sulfates are found in all body cells, with connective tissues, bone, and cartilage having the largest quantities.
Related articles
1. How many electrons are in sulfur?
2. Sulfur valence electrons
3. Mechanical energy examples
4. High yield bonds
5. Chemical reaction definition
6. Chlorella powder
