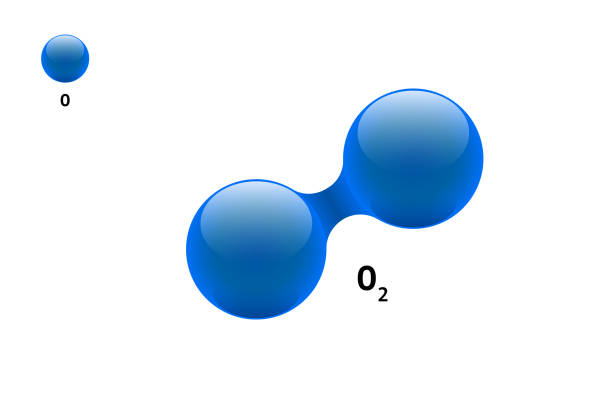
Is o2 Polar? It’s a nonpolar covalent bond because neither particle exerts more force than the other. The 4 electrons that make up the double bond between the two oxygen atoms in diatomic Oxygen are shared evenly between the two elements.
 Is o2 Polar?
Is o2 Polar?
Two oxygen atoms in diatomic Oxygen share the four electrons that make up the double bond. Each element has no partial charges because they are all electronegative. Nonpolar covalent bonds are formed when neither atom exerts more force.
O is the atomic number and symbol for Oxygen, a chemical element. One of the most reactive non-metallic substances belongs to group 16 of the periodic table (the chalcogen group).
This oxidizing agent can quickly oxidize multiple substances. O2 is the most stable of the oxygen allotropes discovered to date. In addition to diatomic Oxygen and molecular Oxygen, it is also known as atmospheric Oxygen.
| Atomic Number | 8 |
| Melting Point | −218.4 °C (−361.1 °F) |
| Boiling Point | −183.0 °C (−297.4 °F) |
| Density (1 atm, 0 °C) | 1.429 g/litre |
| Oxidation States | −1, −2, +2 (with fluorine) |
| Electron Configuration | 1s 22s 22p 4 |
 The Fundamental Principle Of Polarity
The Fundamental Principle Of Polarity
The negative and positive charge centers do not coincide. A polar bond can only form when two atoms are involved, and one of them must be more electronegative than the other to draw some negative charge on itself.
Consequently, the less electronegative atom retains some positive control, which contributes to the formation of a dipole moment. As a result, polar molecules contain as many of the following:
-
Greater than 0D dipole moment
-
More than 0.4 volts of difference
-
Involvement of the molecule’s two poles in a charge accumulation
-
A wide range of temperature ranges
-
Symmetry is missing
-
Solubility in polar solvents is excellent, while nonpolar solvents are insoluble or sparingly soluble.
-
There’s a good explanation for PCl3’s polarity here.
-
This is how nonpolar molecules differ from polar molecules in terms of their properties:
-
This means that they have a dipole moment of 0.
-
Atoms have a c difference of 0–0.4. (negligible)
-
The poles have no charge buildup.
-
The melting and boiling points are lower.
-
The molecular structure of symmetric molecules
-
You can find out why CS2 doesn’t have a polarity here.
As a result, when determining the polarity of any molecule or element, these qualities can be compared for a more precise conclusion.
Summary
Because neither atom nor molecule exerts more force than the other, this is a nonpolar covalent bond. In diatomic Oxygen, the two oxygen atoms share their four electrons equally to form a double bond.
 O2’s Non-Polarity
O2’s Non-Polarity
Due to the following properties, O2 is considered a nonpolar molecule. To determine the polarity of a chemical, examine the following points.
 Differences In Electronegativity
Differences In Electronegativity
A single oxygen atom (O) has an electronegativity of 3.44. When two atoms form a double bond, it creates the molecular O2 structure (diatomic Oxygen). There is no change in the electronegativity between the two atoms.
 O2 Dipole Moment
O2 Dipole Moment
Each atom has an equal and opposing action that makes up O2. The net force on the shared electrons is zero since the magnitude of the draw exerted on the electrons is equal on both sides. Because there is no charge buildup at any pole, the molecular Oxygen’s net dipole moment remains 0 Debye.
 Asymmetrical Shape
Asymmetrical Shape
Because of the diatomic molecule, the O2 molecule has a linear form. Two atoms establish double bonds to produce a linear geometric structure to complete their octet.
 BO2’s Bonding Structure
BO2’s Bonding Structure
The net charge on the atoms in the molecule is zero since there is an equal sharing of electrons and no partial payments. A covalent double bond is formed when two oxygen atoms share one pair of electrons. Thus, the diatomic oxygen molecule (O2) is nonpolar because of its covalent link and zero permanent dipole moment.
Properties
| It is a tasteless, odorless gas. | |
| Liquid at room temperature. | |
| Most elements save noble gases, produce oxides with them. | |
| Oxygen is highly paramagnetic. | |
| In allotropic forms (monoatomic, diatomic, triatomic). | |
| It aids combustion. |
Summary
Whenever two oxygen atoms come together, they form a covalent double bond, in which the valence electrons are evenly distributed across the two atoms. Oxygen’s electron structure is [He] 2s2 2p4. In the second shell, six valence electrons can be used for bonding.
 O2’s Lewis structure
O2’s Lewis structure
A single and double bond can be found in the Lewis structure of diatomic oxygen molecules, but which one is more likely?
However, only one of these two structures is solid enough to remain in place for long periods. Although structures (a) and (b) have total octets for both oxygen atoms, the two configurations differ significantly in terms of stability.
In structure (a), both oxygen atoms have an unpaired electron, called a radical. This radical is very reactive and causes the entire structure to become unstable.
In contrast, there is no radical in structure (b). For structural stability, double bonds are formed, which include each of the radicals in structure (a) in their entirety.
The structure of any element should focus not only on the completion of the octet but also on the system’s stability. O2 elemental Oxygen has two oxygen atoms. Hence the preferable structure is (b) with a double bond connecting the two oxygen atoms.
 O2 - Oxygen’s Stablest Allotrope
O2 - Oxygen’s Stablest Allotrope
It would be best to study the O2 Lewis Structure, Molecular Geometry, and Hybridization. Oxygen exists in the most stable form as O2. It is colorless, but it imparts a soft blue hue in Liquid and solid states.
The process of photosynthesis is primarily responsible for the creation of O2, Atmospheric Carbon Dioxide (Carbon dioxide), and water are combined to produce glucose and Oxygen (Dioxygen)
 Occurrence
Occurrence
Oxygen is the most abundant chemical element on the Earth’s surface, and the third most plentiful feature in the entire universe, in terms of total mass (after Hydrogen earth sum). The group accounts for around 49.2 percent of the Earth’s crust and 0.9 percent of the Sun’s mass.
Oxides, such as silicon dioxide, are the most common forms of Oxygen (SiO2). The Earth’s found in water and the atmosphere in a dissolved state. Oxygen accounts for 88.8% of the mass of the oceans throughout the planet and 20.8 percent of the volume of the atmosphere.
 O2’s Physical and Chemical Characteristics
O2’s Physical and Chemical Characteristics
-
(molality) = 16.6
-
The gaseous density is 1.43 g/L at STP.
-
The density is 1.14 g/cm3
-
At -218.8 °C, the melting point is
-
-183 °C is the boiling point.
-
Oxygen is a colorless gas at STP, but it takes on a pale blue hue when it becomes a liquid.
-
Reactive nonmetal with fusion heat 0.44kJ/mol and vaporization heat 6.82kJ/mol is highly receptive.
-
Pure Oxygen is heavier than air by 1.1 times.
-
There are three distinct oxidation states of Oxygen: -1, -2, and +2. (only in compounds with Fluorine).
-
Water quickly dissolves oxygen (more readily in freshwater than seawater).
-
Coal is combustible because it relies on oxygen for fuel.
 O2’s Various Applications
O2’s Various Applications
Existence and respiration are essential for the survival of all living organisms, whether on land, in the air, or water. Fish and other aquatic species have access to it since it dissolves in water.
In the medical field, oxygen supplementation is also employed. Therapy with oxygen can treat several conditions, including pneumonia and heart disease.
Astronauts use it as a low-pressure breathing gas in space suits. Additionally, scuba and other underwater divers use cylinders to inhale this gas intentionally.
Iron ore smelting for steel production consumes almost half of all commercially producedOxygenn. Welding and water treatment plants also make use of this material. Oxygen is used in several chemical reactions to produce oxides, polymers, etc.
 Formation Of Ozone (O3)
Formation Of Ozone (O3)
Earth’s atmosphere contains ozone, a gaseous component that shields the planet from the Sun’s harmful UV radiation. Two mol of O3 can be formed via an endothermic reaction on three mol of O2 ——–> 2O3 (Oxygen) (Ozone).
Summary
As O2, Oxygen is a two-atom molecule. There are two kinds of atoms. Both particles have the same electronegativity that both fractions have the same effect. The O2 molecule has a zero dipole moment, which indicates that it is nonpolar.
 Commercial Output And Consumption
Commercial Output And Consumption
When large quantities of oxygen are needed, liquid air is fractionally distilled. Nitrogen and argon have lower boiling points thanOxygenn. Hence they are less prone to evaporation. Compressed gas expands and cools as it grows in this process.
The following are significant steps in the procedure:
-
Particulate matter is removed from the air using a filtering process.
-
The alkali absorbs carbon dioxide and water vapor.
-
Standard cooling processes are used to reduce the volume of air and eliminate the heat generated during compression;
-
Air is pushed through coils in a chamber that has been compressed and chilled.
-
The coils are cooled by allowing some compressed air (about 200 atmospheres of pressure) to expand in the chamber.
-
It is then returned to the compressor for further expansion and compression until the compressed air is liquefied at approximately 196 degrees Fahrenheit.
-
The rare light gases are first distilled out of the liquid air, followed by the nitrogen, which leaves the liquid oxygen.
-
Most industrial applications require a product that is 99.5 percent pure.
Summary
The use of pure oxygen in the incineration of trash in closed systems has grown in importance. Liquefied oxygen (LOX) is the oxidizer fuel for rocket oxidizer fuels; the amount of LOX consumed depends on the amount of activity in space.
 Oxygen And Our Environment
Oxygen And Our Environment
The Earth’s crust comprises silicon-oxygen rocks, but there are also oxides of numerous other elements. More than a million trillion metric tonnes of oxyEarth’s composes one-fifth of the atmosphere.
The photosynthesis of plants releases oxygen into the atmosphere, which has built up over a long period due to the abundance of carbon dioxide present in the early environment.
In rivers, lakes, and the world’s oceans, life is conceivable because oxygen is easily dissolved in water. When the oxygen supply in rivers and lakes is diminished, fish and other aquatic creatures will no longer survive.
Oxygen binds to nearly every chemical except for inert gases. Among the most common binary, Oxygen compounds, water, and silica, the primary component of sand.
Most rocks and soils are composed of silicates, the most common compound with more than two elements. As a source of the metals, aluminum oxide (bauxite) and iron oxides are common in nature, as well as calcium carbonate (limestone and marble) and calcium sulfate (gypsum).
 Oxygen’s Health Benefits
Oxygen’s Health Benefits
As a component of DNA and nearly every other physiologically significant substance, Oxygen is fundamental to all life. For the sake of survival, animals must have a constant supply of gas at all times.
The iron atom at the center of hemoglobin in the blood picks up oxygen from the lungs transports it to where it is needed. Oxygen is essential for life, but too much can be harmful, as with many other things.
Lung injury can develop if exposed to high Oxygen levels for a lengthy period. Lung injury is caused by inhaling 50 to 100 percent oxygen at normal pressure for a protracted period.
Lung function tests should be performed on people who operate in environments where they are regularly exposed to high concentrations of pure oxygen. You should wear specific clothing to keep your body from freezing because oxygen is typically stored at low temperatures.
 How Does Oxygen In Water Affect Our Health?
How Does Oxygen In Water Affect Our Health?
It is estimated that 60 percent of the human body’s weight is oxygen. Its value can fluctuate significantly because it is primarily found in water molecules. Humans take in oxygen through their lungs, which are then transported to numerous organs via the blood.
A network of tiny capillaries transports the substance. There must be a 6-8 mg/L concentration for this to work. Cancer and cardiovascular disease are linked to the production of oxygen radicals. Asphyxiation occurs when the oxygen concentration in the airdrops is below 3%.
At a concentration of less than 7%, a person is at risk of losing consciousness. It’s possible that breathing in too much oxygen will kill you. Cramping is a common occurrence for sports divers who use pure oxygen. Infants given too much oxygen in incubators are likely to go blind. Oxygen as ozone can harm the lungs, so be careful.
Hyper oxides, peroxides, and hydroxyl radicals are some of the most toxic forms of oxygen. “Blowing” high carbon steel—volatilizing carbon dioxide and other nonmetal impurities—is the primary use of pure oxygen in the steel industry, which is a quicker and more easily regulated process than if the air were utilized.
Summary
Hydroxyl, carbonyl, and other functional groups include oxygen atoms. Hemoglobin binds to it in the blood and then stores it in muscles as myoglobin. Drinking water that contains oxygen aids in the creation of a protective layer on the inside of metal water pipes.
 Frequently Asked Questions - FAQs
Frequently Asked Questions - FAQs
These are the most frequently asked Questions about o2 polarity,
 Is o2 a polar or a nonpolar molecule, and why?
Is o2 a polar or a nonpolar molecule, and why?
Bonds between atoms of the same element or distinct elements that share electrons evenly are called nonpolar covalent bonds. Molecular Oxygen (O2) is an example of a nonpolar molecule because the electrons are evenly distributed among the two oxygen atoms.
 Is O2 polar or charged?
Is O2 polar or charged?
Phenols (3.5) and Alcohols (3.5). Carbon-oxygen and hydrogen-oxygen bonds are polar because oxygen atoms are more electronegative than carbon or hydrogen atoms. Carbon and hydrogen atoms have a little optimistic and a slightly negative charge, respectively.
 Is Oxygen polar?
Is Oxygen polar?
In this case, the two bonds created will be polar apart nearly every chemical, almost every chemical ent, Oxygen has a partial negative charge. In contrast, the two less electronegative atoms, hydrogen, have two partial positive accounts.
 What is O2’s molecular structure?
What is O2’s molecular structure?
That molecular shape and bond angles of 1 silicates because both oxygen atoms in the molecule have identical electronegativity, and both atoms share equal proportions of bound shared electrons.
 Is O bond polar or nonpolar?
Is O bond polar or nonpolar?
Polar covalent links can generate a dipole moment in molecules with one or more polar covalent bonds. Because hydrogen and oxygen have varying electronegativities, the O-H covalent bond in water is polar.
 In what ways does oxygen influence polarity?
In what ways does oxygen influence polarity?
Because of its high electronegativity, Oxygen forms polar connections with other elements. it is commonly used as an oxidizing reagent.
 What is the molecular formula for oxygen?
What is the molecular formula for oxygen?
Two atoms of the element create dioxygen, a colorless and odorless gas with O2 and pressure. Earth, in so many cases’ atmosphere, contains 20.95 percent diatomic oxygen gas. However, this has fluctuated dramatically over extended periods.
 What is a healthy level of oxygen?
What is a healthy level of oxygen?
A healthy person’s blood oxygen level is typically 95% and 100%. As a result, the vast majority of your red blood cells transport oxygen to your cells and tissues.
 What makes O2 a gas?
What makes O2 a gas?
In nature, Oxygen is found as a molecule, not as a metal. As you can see, each one has two oxygen molecules attached by a powerful bond. Oxygen is a gas at standard temperature because it has a low melting and boiling temperatures.
Conclusion
Oxygen in sewage treatment holds very fine capillaries and delivers it more efficiently than other chemical procedures now in service. To produce oxidation-controlled compounds like acetylene,thylssubsequently stored in myoglobin muscles-enriched air has substituted standard atmosphere.
Oxygen tents, inhalators, and pediatric incubators are examples of medical uses for oxygen. During general anesthesia, oxygen-enriched gaseous anesthetics keep the patient alive. Several businesses that employ kilns requireOxygenn.
 Related Articles
Related Articles
1 - Oxygen atom
2 - Test for oxygen
3 - Oxygen formula